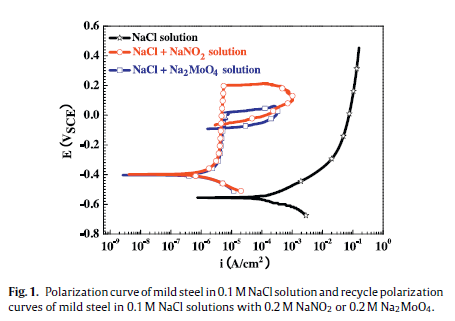
The inhibitive mechani** of NO2−and MoO42−on the initiation and propagation of pitting corrosion formild steel in chloride solution was studied with electrochemical methods and X-ray photoelectron spec-troscopy (XPS). In 0.1 M NaCl solution both the addition of 0.2 M NaNO2and 0.2 M Na2MoO4effectivelypromoted passivation of mild steel. The passive film on the steel surface formed in NaCl + NO2−solutionwas composed of mainly _-Fe2O3, and the film formed in NaCl + MoO42−solution was composed of twocomponents: one is Fe2(MoO4)3and the other is an oxide composed of Fe and O. The film formed in NaNO2solution has lower oxygen vacancies and larger impedance than the film formed in Na2MoO4solution.NO2−shows better inhibition to the initiation of pitting corrosion than MoO42−, which is attributed toits strong oxidability that results in the formation of a stable _-Fe2O3film. However, in NaNO2solution,once a pit forms, it is more difficult to get repassivated than the situation in Na2MoO4solution. Themain reason is due to that in a propagating pit MoO42−anions result in increased solution pH value, butconversely NO2−anions lead to a decreased solution pH value within a pit.